Researchers in the labs of UC Santa Barbara materials assistant professor Chris Bates and professor and department chair, Michael Chabinyc, have teamed up to develop the first 3D-printable “bottlebrush” elastomer. The new material results in printed objects that have unusual softness and elasticity — mechanical properties that closely resemble those of human tissue.
Conventional elastomers, i.e. rubbers, are stiffer than many biological tissues. The reason is related to the size and shape of their constituent polymers, which are long, linear molecules that easily entangle, like a bowl of cooked spaghetti. In contrast, bottlebrush polymers have additional polymers attached to the linear backbone, leading to a structure that is more akin to a bottle brush you might find in your kitchen. The bottlebrush polymer structure imparts the ability to form extremely soft elastomers.
The ability to 3D-print bottlebrush elastomers makes it possible to leverage these unique mechanical properties in applications that require careful control over the dimensions of objects ranging from biomimetic tissue to high-sensitivity electronic devices, such as touch pads, sensors, and actuators.
Two postdoctoral researchers — Renxuan Xie and Sanjoy Mukherjee — played key roles in developing the new material. Their findings were published in the Nov. 13 issue of the journal Science Advances.
Xie and Mukherjee’s key discovery involves the self-assembly of bottlebrush polymers at the nanometer length scale, which causes a solid-to-liquid transition in response to applied pressure. This material is categorized as a yield-stress fluid, meaning it begins as a semi-soft solid that holds its shape, like butter or toothpaste, but when sufficient pressure is applied, it liquefies and can be squeezed through a syringe. The team exploits this property to create inks in a 3D-printing process called direct ink writing (DIW).
The researchers can tune the material to flow under various amounts of pressure to match the desired processing conditions. “For instance, maybe you want the polymer to hold its shape under a different level of stress, such as when vibration is present,” says Xie. “Our material can hold its shape for hours. That’s important, because if the material sags during printing, the printed part will have poor structural stability.”
Once the object is printed, UV light is shined onto it to activate crosslinkers that Mukherjee synthesized and included as a part of the ink formulation. The crosslinkers can link up nearby bottlebrush polymers, resulting in a super-soft elastomer. At that point, the material becomes a permanent solid — it will no longer liquefy under pressure — and exhibits extraordinary properties.
“We start with long polymers that are not crosslinked,” says Xie. “That allows them to flow like a fluid. But, after you shine the light on them, the small molecules between the polymer chains react and are linked together into a network, so you have a solid, an elastomer that, when stretched, will return to its original shape.”
The softness of a material is measured in terms of its modulus, and most elastomers have a rather high modulus, meaning their stiffness and elasticity are similar to those of a rubber band. “The modulus of our material is a thousand times smaller than that of a rubber band,” Xie notes. “It is super-soft — it feels very much like human tissue — and very stretchy. It can stretch about three to four times its length.”
An Accidental Ink
Mukherjee discovered the material by accident while trying to develop a material for a different project, one that would increase the amount of charge that can be stored by an actuator. When the elastomer came to Xie for characterization, he knew immediately that it was special. “I could see right away that it was different, because it could hold its shape so well,” he recalls.
“When we saw this really well-defined yield stress, it dawned on everyone collectively that we could 3D-print it,” Bates says, “and that would be cool, because none of the 3D-printable materials we know of have this super-soft property.”
Bottlebrush polymers have been around for more than twenty years, but, Bates says, “The field has exploded in the past ten years thanks to advances in synthetic chemistry that provide exquisite control over the size and shape of these unique molecules. “These super-soft elastomers might be applicable as implants,” he adds. “You may be able to reduce inflammation and rejection by the body if the mechanical properties of an implant match native tissue.”
Another important element of the new material is that it is pure polymer, Chabinyc notes. “There’s no water or other solvent in them to artificially make them softer.”
To understand the importance of having no water in the polymer, it’s helpful to think of Jell-O, which is mostly water and can hold its shape, but only as long as the water remains inside. “If the water went away, then you’d just have a shapeless pile of material,” says Chabinyc. “With a conventional polymer, you must figure out how to keep the right amount of water in it to maintain its structure, but this new material is all solid, so it will never change.”
Moreover, the new material can be 3D-printed and processed without solvent, which is also unusual. “People often add solvent to liquify a solid so that it can be squeezed out of a nozzle,” says Xie, “but if you add solvent, it has to evaporate after printing, causing the object to change its shape or crack.”
Mukherjee adds, “We wanted the material and the printing process to be as clean and as easy as possible, so we played a chemistry trick with solubility and self-assembly, which enabled the solvent-free process. The fact that we don’t use solvent is a tremendous advantage.”
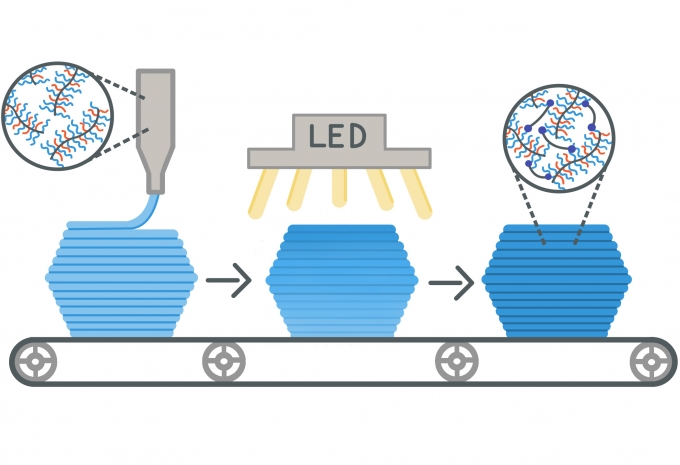
From left: the unlinked polymer ink, infrared light being applied to activate the crosslinks, and the final product — a super-soft, super-elastic crosslinked elastomer. Illustration by Isabelle Chabinyc
